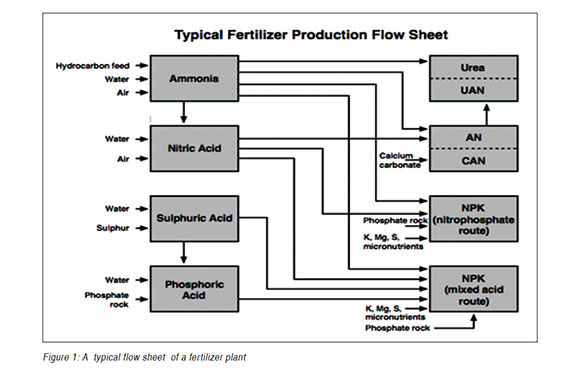
Fertilizers
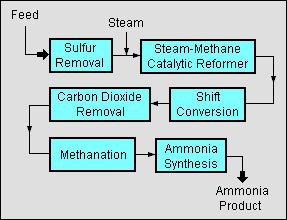
The primary industrial method for ammonia synthesis is the Haber-Bosch process, created by Fritz Haber in 1905 and developed for industry by Carl Bosch in 1910. The overall process synthesizes ammonia from molecular Nitrogen (N2) and Hydrogen (H2) by feeding the reactants over iron catalysts at a high pressure and temperature. The H2 is first produced by one of the syngas production processes described previously, and then separated into a pure H2 stream. The N2 and H2 are reacted in a ratio of 1:3 in the following reaction:
N2 + 3H2 → 2NH3
The ammonia product is recovered in a liquid or gaseous form, depending on whether it will be sold as is or converted further to other products.
For urea production, the ammonia is then reacted with CO2 (which could be provided from the AGR system) in the following two-step reactions at high temperature and pressure:
2NH3 + CO2 ↔ NH2COONH4 (ammonium carbamate)
NH2COONH4 ↔ H2O + NH2CONH2 (urea)
The urea contains a mixture of unreacted NH3 and CO2. As the pressure is reduced and heat is applied, the ammonium carbamate decomposes to NH3 and CO2, which are recycled to produce more ammonium carbamate. The urea solution is concentrated and then granulated for use as fertilizer.