Partial Oxidation
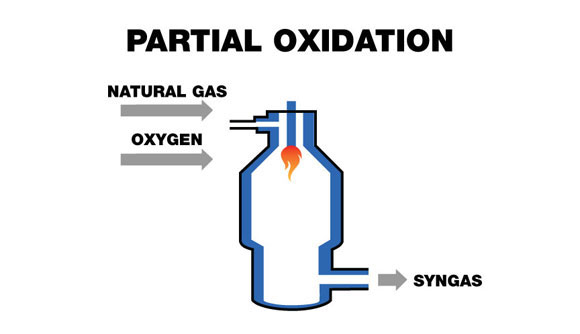
Partial oxidation (POX), or gasification, is a chemical reaction that occurs when a mixture of a hydrocarbon feedstock and a sub-stoichiometric amount of pure oxygen (O2) are reacted together, producing a syngas stream with a typical H2/CO ratio range of 1.6 to 1.8.
The hydrocarbon feedstock is fed into the POX reactor (see figure below), where the carbon in the feedstock is reacted with oxygen in an exothermic reaction, forming carbon monoxide (CO). Since there is a lack of oxygen, the reaction does not complete to form carbon dioxide (CO2).
C + ½ O2 → CO
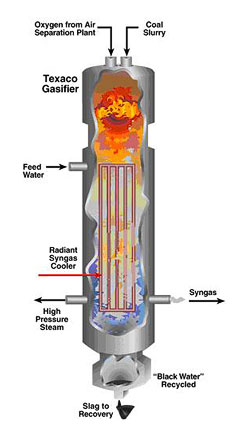
Below is a schematic of a GE/Texaco Gasifier for coal gasification
Typically, all or a portion of the CO then flows to a water shift reactor, where the CO reacts with steam, forming a mixture of CO and H2:
There are two primary types of POX systems: 1) Thermal POX (TPOX), which occurs at >2200°F and is used with high sulfur feedstocks, and 2) Catalytic POX (CPOX), which uses low sulfur feedstocks with a sulfur-sensitive catalyst, allowing the reactions to occur in a lower temperature range of 1475-1650°F, which reduces energy consumption.
CO + H2O → CO2 + H2
The first reaction is the reforming reaction, and the second reaction is the water gas shift reaction. Both reactions produce hydrogen and are both limited by thermodynamic equilibrium. The exit equilibrium temperature from the reformer is typically in the range of 1500°F to 1700°F. The net reaction is highly endothermic, requiring significant heat. These reactions take place under carefully controlled external firing within the reformer furnace.


