Steam Methane Reforming (SMR): Process, Technology & Industrial Applications
Steam methane reforming (SMR) is one of the most important production pathways within GSTC’s broader syngas technology landscape.
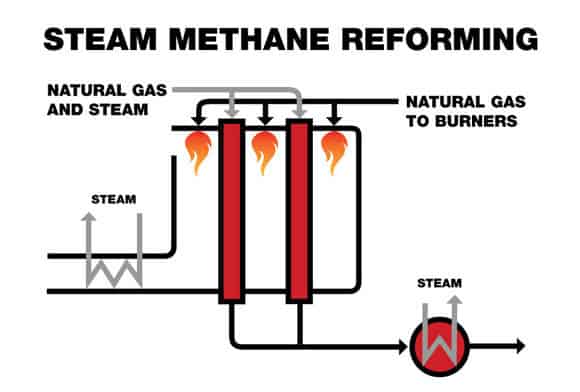
What is Steam Methane Reforming?
Steam methane reforming (SMR) is the dominant industrial method for hydrogen production from natural gas. It converts natural gas (mainly methane) and steam into hydrogen, carbon monoxide, and carbon dioxide in a high-temperature catalytic process.
SMR is widely used because it has been proven at an industrial scale, it is highly reliable, it integrates well with existing natural gas infrastructure, and it supports large-scale hydrogen demand across refining, ammonia, and chemical production.
Steam Methane Reforming Reaction & Chemical Equations
Steam methane reforming proceeds through two main reaction stages. Methane acts as both a reactant and as the fuel that supplies the heat required for the reaction. First, methane reacts with steam in the reformer. Then the resulting gas passes through the Water-Gas Shift stage to increase the hydrogen yield.
Step 1: Steam Methane Reforming Reaction
CH4 + H2O ⇌ CO + 3H2
This is the core SMR equation. It shows methane reacting with steam to produce syngas, that is, carbon monoxide and hydrogen.
Step 2: Water-Gas Shift Reaction
CO + H2O ⇌ CO2 + H2
This secondary reaction increases the total hydrogen yield by converting carbon monoxide to carbon dioxide and additional hydrogen.
Overall Reaction
CH4 + 2H2O ⇌ CO2 + 4H2
This overall expression is often used to describe hydrogen production from natural gas through steam reforming.
Carbon dioxide and hydrogen are easily separated and the CO2 may be sequestered by well proven processes.
Steam Methane Reforming Process (Step-by-Step)
The steam methane reforming process converts natural gas into high-purity hydrogen through a sequence of integrated unit operations. It is designed for reliability, catalyst protection, thermal efficiency, and large-scale industrial performance.
Step 1: Feedstock Preparation (Natural Gas Treatment)
Before reforming begins, natural gas must be treated to remove sulfur compounds that can damage the catalyst.
Input → Treatment → Output
- Raw natural gas containing sulfur species
- Hydrodesulfurization and zinc oxide adsorption
- Clean, sulfur-free methane suitable for reforming
Even trace sulfur can poison a nickel catalyst. Effective desulfurization is therefore essential for stable performance and catalyst life.
Step 2: Reforming Reactor (High-Temperature Reaction)
The steam methane reformer is the core system in the plant. A mixture of methane and steam flows through catalyst-filled tubes located inside a fired furnace. External burners provide the heat needed to sustain the reaction, while the nickel-based catalyst promotes methane conversion.
Typical operating temperatures are in the range of 1500–1700°F (815–925°C). Reactor design, heat transfer, and steam ratio all influence conversion, fuel use, and plant economics.
Step 3: Water-Gas Shift Reaction
After the reformer, the gas stream undergoes Water Gas Shift to increase hydrogen yield.
CO + H2O ⇌ CO2 + H2
This step converts carbon monoxide into additional hydrogen and reduces CO ahead of purification. Industrial systems typically use both high-temperature and low-temperature shift reactors to optimize performance.
Step 4: Hydrogen Purification (PSA)
The final hydrogen-rich stream is commonly purified using Pressure Swing Adsorption (PSA). In PSA, impurities such as CO2, CO, methane, and trace gases are selectively adsorbed under pressure. The beds are then regenerated through pressure cycling.
The result is hydrogen purity that typically exceeds 99.9%, depending on plant design and product specification.
Looking to understand how SMR fits into real-world projects and investment decisions?
GSTC connects technical insight with industry context, linking hydrogen production pathways such as SMR to project development, partner selection, and execution strategy. Learn more about our mission.
Steam Methane Reforming Efficiency & Performance
The efficiency of steam methane reforming typically ranges from 65% to 85%, depending on plant configuration, thermal integration, operating conditions, and downstream recovery.
What drives SMR efficiency?
- Heat recovery from reformer flue gas
- Desired steam-to-carbon ratio
- Reformer furnace and tube performance
- Shift conversion efficiency
- PSA recovery performance
SMR is energy-intensive because the reforming reaction requires continuous external heat. Higher-efficiency plants typically include more extensive heat recovery and integration, but that improvement often comes with higher capital cost.
From a project standpoint, SMR performance is typically evaluated as a balance among efficiency, reliability, fuel costs, and capital intensity.
Most professionals evaluating SMR do not start from scratch; they validate assumptions. At the Global Syngas Technologies Council, you get access to technical perspectives, project data, and peer-level discussions that help decision-makers move from theory to confidence.
Applications of Steam Methane Reforming
Steam methane reforming underpins a wide range of industrial hydrogen applications. Its role extends across energy, chemicals, and manufacturing sectors.
- Fertilizers: Hydrogen for ammonia synthesis
- Refining: Hydrogen for refinery hydro-processing for cleaner fuel production
- Chemicals: syngas for methanol synthesis and its family of chemical derivatives
- Energy: Syngas and hydrogen for power plant fuel
Advantages and Disadvantages of Steam Methane Reforming
Advantages of SMR
- Proven reliability at an industrial scale
- Widely used for syngas and hydrogen production from natural gas
- Strong integration with existing infrastructure
- Suitable for high-volume, continuous hydrogen demand
- Established reactor, catalyst, and purification systems
Disadvantages of SMR
- High energy demand
- Significant CO2 emissions without carbon capture
- Dependence on natural gas feedstock
- Requires careful catalyst protection and high-temperature operations
- Faces increasing scrutiny in low-carbon transition planning
The main reason SMR remains widely used is that it combines technical maturity, reliability, scalability, and strong industrial familiarity. Its main challenge is carbon dioxide emissions.
Future of Steam Methane Reforming Technology
The future of steam methane reforming (SMR) will be shaped by lower-carbon hydrogen strategies, improved energy efficiency, and tighter integration with emerging clean energy systems.
Electrified Reforming
Electrified reforming replaces some or all of the fired heat input in a conventional reformer with electric heating. This approach can reduce direct combustion emissions when paired with low-carbon electricity and may improve the carbon profile of hydrogen production.
Carbon Capture Integration
One of the most important developments in SMR is the integration of carbon capture and storage (CCS). By capturing CO₂ from process gas and furnace exhaust, SMR can support lower-carbon hydrogen production, often described as blue hydrogen.
Biomethane as a Feedstock
Another pathway is the use of biomethane in place of conventional natural gas. This allows existing steam reforming infrastructure to remain in service while improving lifecycle carbon performance.
Hybrid Hydrogen Systems
SMR is also being considered as part of hybrid hydrogen systems that combine:
- Steam methane reforming for dependable baseload hydrogen supply
- Electrolysis for flexible, low-carbon hydrogen production
This approach can improve overall system resilience while supporting changing power and fuel markets.
Stay Connected to the Syngas Industry
Explore how SMR, gasification, and hydrogen strategies are being applied across real projects. Subscribe to the GSTC newsletter for updates and upcoming events, or contact our team to discuss membership, technical questions, and opportunities to connect.
FAQs About Steam Methane Reforming
What is steam methane reforming?
Steam methane reforming is the main industrial process used to make hydrogen from natural gas. It uses steam, heat, and a catalyst to convert methane into hydrogen and carbon oxides.
What is the chemical equation of SMR?
The main SMR reaction for syngas production is CH₄ + H₂O ⇌ CO + 3H₂. For hydrogen applications it is followed by the water-gas shift reaction: CO + H₂O ⇌ CO₂ + H₂.
How does steam methane reforming produce hydrogen?
SMR produces hydrogen by reacting methane with steam at high temperature over a catalyst. The gas then goes through shift conversion and purification to increase the hydrogen yield.
What are the operating conditions of SMR?
SMR operates at high temperature, typically around 1500–1700°F (815–925°C). It also requires controlled pressure and steam-to-carbon ratios.
What catalyst is used in SMR?
SMR typically uses a nickel-based catalyst. This catalyst supports methane conversion, but it must be protected from sulfur.
What is the efficiency of steam methane reforming?
SMR efficiency typically ranges from 65% to 85%. Actual efficiency depends on plant design, heat recovery, and purification performance.
Why is SMR widely used for hydrogen production?
SMR is widely used because it is proven, reliable, scalable, and cost-effective when natural gas is available at low cost. It also fits well with existing natural gas and industrial infrastructure.
What are the environmental impacts of SMR?
SMR produces CO₂ emissions from both the chemical reaction and furnace heating. Without carbon capture, it remains a carbon-intensive hydrogen production method.
What is the difference between SMR and electrolysis?
SMR makes hydrogen from natural gas. Electrolysis makes hydrogen from water using electricity.
What are the advantages and disadvantages of steam methane reforming?
The main advantages of SMR are scale, maturity, and cost efficiency. The main disadvantages are CO₂ emissions, high energy demand, and reliance on low cost natural gas.

