Methanation
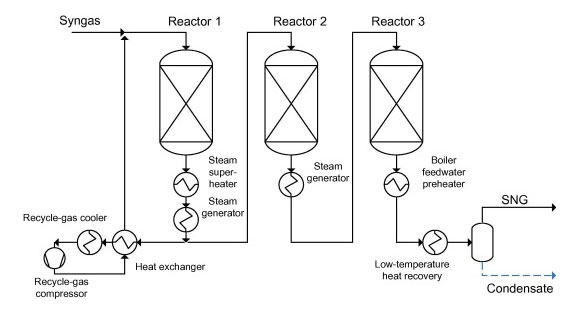
Methanation is the reaction where carbon oxides and hydrogen are converted to methane and water. The methanation reactions are given below:
CO + 3H2 = CH4 + H2O
CO2 + 4H2 = CH4 + 2H2O
The methanation reactions are catalysed by nickel catalysts. In the methanol industry, there are two typical uses for methanation: to purify synthesis gas and to produce methane for substitute natural gas (SNG).

